Design and scenario comparison study of an amine solvent-based carbon-dioxide-capture process for ships
Copyright © The Korean Society of Marine Engineering
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
As the International maritime Organization (IMO) strengthens its greenhouse gas regulations, technologies for capturing CO2 emissions from ships are emerging as a promising short-term option for reducing emissions from existing ships. This study develops an onboard carbon-capture system (OCCS) for a container ship utilizing an amine-based solvent and conducts a comparative analysis of full- and partial-exhaust gas-capture scenarios. Simulations are performed using Aspen HYSYS V14, employing the Acid Gas–Chemical Solvent Property Package. The full-exhaust gas-capture scenario theoretically achieved a high capture efficiency exceeding 90%; however, the requirement for larger absorption and regeneration towers, along with an increased reboiler duty of approximately 3.4 MJ/kgCO2, substantially increased energy consumption. Additionally, the system produced approximately 1,344 tonnes of CO2 for a seven-day voyage, indicating its limited feasibility owing to spatial and weight constraints within the ship environment. Conversely, the partial-exhaust gas-capture scenario was designed to capture 2 tonnes of CO2 per hour with an Liquid flow/Gas flowL/G ratio range of 2.5. The reboiler duty was approximately 2.9 MJ/kgCO2, reflecting an approximately 15% lower energy consumption compared to the full-exhaust capture scenario. Furthermore, the storage-tank-capacity requirement was approximately 425 m3, which was approximately 25% of that required for the full-exhaust capture scenario. This significantly alleviated the spatial burden when applied to ships. Consequently, this study suggests that partial-exhaust gas capture is technically and economically viable in the initial commercialization phase of marine OCCS, and a phased introduction strategy culminating in full-exhaust capture as a long-term objective is desirable. These findings can serve as foundational data for future marine OCCS designs and International Maritime Organization regulatory response strategies.
Keywords:
Onboard Carbon-Capture System (OCCS), Carbon capture, Carbon-Intensity Indicator (CII), Monoethanolamine (MEA), Process Simulation1. Introduction
The International Maritime Organization (IMO) is gradually strengthening regulations to reduce greenhouse-gas emissions in the shipping sector. Specifically, the Energy Efficiency Design Index (EEDI), Energy-Efficiency Existing Ships Index (EEXI), and Carbon-Intensity Indicator (CII) directly limit the fuel efficiency and carbon-dioxide emissions of both new and existing ships. The IMO aims to reduce emissions by 40% by 2030 compared with 2008 levels and achieve net-zero emissions in the shipping sector by 2050 [1][2]. Furthermore, a specific roadmap to achieve these reduction goals was adopted at the 80th Marine Environment-Protection Committee (MPEC) meeting, which significantly impacted ship design and operations [3].
Onboard carbon-capture system (OCCS) technology for ships is attracting attention as a realistic alternative because of its higher technological maturity compared to conventional fuel conversion (e.g., ammonia and hydrogen) and its potential for retrofitting in existing ships [4]-[7]. According to a DNV (Det Norske Veritas) report, achieving carbon neutrality by 2050 requires the diversification of alternative fuels as well as the adoption of direct carbon-capture technologies, such as OCCS [8][9].
Capture technologies utilizing amine-based solvents have already been demonstrated in onshore power plants and oil and gas plants, and high capture efficiencies have been reported in studies applying them to ships. For example, Luo and Wang [5] demonstrated that capture efficiencies exceeding 90% could be achieved using a monoethanolamine (MEA)-based absorption process, and Jung and Seo [10] quantitatively analyzed the capture performance using a rate-based model that reflected ship operating conditions. Furthermore, Padurean et al. [11] investigated the thermodynamic behaviors of various amine-solvent systems and suggested their potential for ship applications. Recently, the application of MEA and N-methyldiethanolamine(MDEA)/piperazine (PZ) mixed solvents has been demonstrated to enhance reaction speed and energy efficiency, and stable performance has been verified in MEA/2-amino-2-methyl-1-propanol (AMP) systems under repeated cycles [7][12].
However, practical shipboard applications have several limitations. Key challenges include the size and weight of absorption and regeneration towers, CO2 storage and capacity, and operational safety in the vibration and agitation environments of a ship [4][6]. A techno-economic analysis by Visonà et al. [6] suggested that space constraints and energy loads are key bottlenecks to the feasibility of large container ships; initially, a partial-capture strategy targeting a portion of the exhaust gas, rather than the entire exhaust gas, may be feasible.
Recently, active research has been conducted on the economic feasibility and environmental impact of ship-based OCCS. The trade-off between the L/G ratio and energy consumption depending on the vessel size and operating conditions has been systematically analyzed in process-simulation studies, emphasizing the importance of deriving optimal design conditions [5][7]. Furthermore, the results of CCS life-cycle assessments (LCAs) demonstrate that capture technology can substantially contribute to achieving carbon neutrality in the shipping sector [7]. Furthermore, analyses of social acceptability and regulatory aspects highlight the need for a policy framework for the commercialization of ship-based Carbon Capture, Utilization and Storage (CCUS) [4][9].
In the initial stages of commercializing the OCCS, a pragmatic approach is to employ partial capture rather than full capture to comply with IMO regulations while mitigating spatial and economic challenges. This study conducts a comparative analysis of full- and partial-exhaust gas-capture scenarios utilizing an MEA-based CO2-absorption process on a container ship. The objective of this study is to establish design criteria that consider space limitations, equipment dimensions, and energy consumption, which are essential considerations in the design of an OCCS. This study aims to provide foundational data for the development of future commercialization and regulatory response strategies.
Against this background, this study makes the following contributions. First, it develops a detailed, rate-based onboard carbon capture process model for a large container ship and quantitatively compares full and partial-exhaust gas capture configurations under consistent boundary conditions. Second, it explicitly couples absorber/regenerator sizing, Co2 storage capacity, and specific reboiler duty to shipboard space and energy constraints, thereby identifying a practical design envelope for marine OCCS. Third, it proposed a phased deployment strategy in which partial-exhaust gas capture is adopted as an initial compliance option under IMO regulations, with full-exhaust capture defined as a long-term target.
2. Methodology
2.1 Scope of Application
This study aims to evaluate the feasibility of complying with IMO regulations by comparing and analyzing full- and partial-exhaust gas-capture scenarios for a marine carbon-dioxide-capture system applicable to container ships. To achieve this objective, an amine-solvent absorption-based capture process was modeled using Aspen HYSYS V14, a commercial process-simulation tool [5][13].
2.2 Ship and Emissions Information
In this study, a 130,000 DWT container ship was taken as the reference ship. It is equipped with a low-speed, two-stroke dual-fuel main engine, several diesel-driven auxiliary generators, and an auxiliary boiler, and the combined exhaust gas from these units was treated as the main emission source for the OCCS design. The exhaust-gas volume and temperature under each operating condition are presented in Table 1, and the exhaust-gas composition is detailed in Table 2. The representative operating condition was set at a 75% load, reflecting previous reports that the 60%–80% load range is the most frequent in actual commercial ship operations and that the 70%–80% load range accounts for more than 65% of the total voyage, based on the engine-data analysis of large commercial ships [4][8]. Furthermore, the ship considered in this study was assumed to be equipped with an SOx scrubber so that most of the SOx and a substantial portion of the particulate matter and other impurities were removed upstream of the CO2-capture unit [14][15].
In this study, the composition of the gas entering the capture process was simplified into four components: N2, O2, H2O, and CO2. This simplification was implemented to enhance the safety of the simulations and concentrate on the thermodynamic and reaction analyses of the capture process. However, in actual operational environments, trace amounts of NOx and SOx may persist, potentially leading to solvent degradation and scaling issues during prolonged operation [7][10].
2.3 Reaction Mechanism of Amine Solvent
This study employed an aqueous MEA solution, which is the most extensively used amine solvent. MEA is characterized by a rapid reaction rate and high capture efficiency, and has been widely adopted and validated in existing power and petrochemical plants [10][16]. Previous research has demonstrated that MEA exhibits excellent reactivity with CO2 because of its low molecular weight and high hydrophilicity, making it the most promising amine solvent for onboard carbon-capture applications [7][12].
The primary reactions between MEA and CO2 are summarized as follows [17]:
| (1) |
| (2) |
| (3) |
| (4) |
| (5) |
| (6) |
| (7) |
Equation (1) represents the water-dissociation reaction, which supplies the initial OH- necessary for CO2 absorption. Equation (2) describes the equilibrium between HCO3- (bicarbonate) and CO3-2, which determines the buffering capacity of the solution. Equation (3) describes the protonation/deprotonation equilibrium of MEA, which regulates the effective base strength of the solvent. Equation (4) shows the reaction between OH- and CO2, a significant pathway during the initial absorption phase. Equation (5) details the bicarbonate dissociation reaction, providing the mechanism for CO2 re-release, whereas Equation (6) describes the formation of MEACOO- (carbamate), accounting for a substantial portion of the total CO2 absorption. Equation (7) describes the decomposition of carbamate, which facilitates CO2 release during the regeneration stage and is crucial for determining the energy requirements for regeneration. Among these reactions, carbamate formation and decomposition constitute the main regeneration cycle that governs the specific reboiler duty and largely determines the required size of the regenerator and associated heat-exchange equipment in the OCCS. These reaction mechanisms and kinetics were initially introduced in the seminal work of Caplow [17] and were later systematically summarized in a comprehensive review of CCS by Bui et al. [7]. Additionally, a recent study by Said et al. offered an extensive explanation of the various amine-CO2 reaction pathways, supporting the design feasibility of OCCS processes for maritime applications [18]. Consequently, this reaction is a critical factor that directly affects the economic viability of the OCCS process.
2.4 Simulation Software
The simulations conducted in this study utilized Aspen HYSYS V14, which employs the Acid Gas-Chemical Solvent Property Package. This package is specifically designed for CO2-removal processes using amine solvents and is capable of accurately predicting the behavior of amine-CO2 systems. This is achieved by simultaneously considering chemical equilibrium, the reaction rates, and electrolyte equilibrium. The model calculates non-ideal mixing behavior based on the electrolyte non-random two-liquid(eNRTL) model and has been validated in industrial settings, such as power plants and natural-gas-processing facilities [13].
2.5 Process Configuration
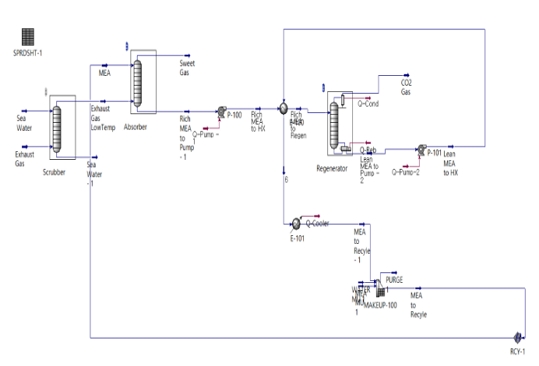
Figure 1 depicts the OCCS process modeled in this study, which represents the most commonly employed wet amine absorption-regeneration process. The entire system comprises an absorption tower, regeneration tower, heat exchanger, pump, and make-up line. This configuration has been validated extensively in onshore power and gas plants. When applied to maritime vessels, it is designed to accommodate operational constraints while maintaining the fundamental flow structure [5][6][12].
Initially, exhaust gas cooled to approximately 40 ℃ is introduced at the base of the absorption tower. Concurrently, an amine solvent is introduced at the top, facilitating contact between the downward-flowing solvent and upward-flowing exhaust gas, thereby initiating a CO2-absorption reaction. During this interaction, the primary component, MEA, reacts with CO2, forming carbamate or bicarbonate. The resulting sweet gas, devoid of CO2, is expelled from the top of the absorption tower.
Subsequently, the rich amines are extracted from the bottom of the absorption tower. This solution is pressurized to approximately 4 bar using a pump and subsequently heated to approximately 120 ℃ via a heat exchanger. This heat-exchange process is crucial for optimizing the energy efficiency of the absorption-regeneration cycle by increasing the temperature of the rich amine prior to its entry into the regeneration tower.
In the next phase, the rich amine introduced at the top of the regeneration tower undergoes separation from CO2 upon receiving heat at approximately 140 ℃ from a reboiler. The high-purity CO2 produced during this process is captured at the top of the tower and conveyed to an onboard CO2 liquefaction and storage system. Concurrently, the lean amine, from which the CO2 has been removed, is discharged from the bottom of the regeneration tower.
Subsequently, the discharged lean amine traverses a heat exchanger to transfer heat to the rich amine, followed by passage through a cooler to adjust its temperature to approximately 40 ℃. It is then recycled and reintroduced to the top of the absorption tower, thereby completing the cycle.
Finally, the system incorporates a make-up line. During operation, a portion of the amine solvent is lost due to deterioration, volatilization, and byproduct reactions. Consequently, fresh solvent is replenished through the make-up line to maintain process stability [5][10][13]-[19]. The simulation outputs were analyzed using descriptive statistics, and key performance indicators such as CO2 capture rate, specific reboiler duty, and required storage volume were compared as absolute vales and percentage differences between the full and partial-exhaust gas capture scenarios.
3. Results and Discussion
3.1 Full-Exhaust Gas-Absorption Scenario
A simulation was conducted in which the entire exhaust gas was processed through the absorption tower, and the absorption efficiency was maintained at 90%. The tower height was set at 7.5 m, considering that it is approximately 10 m, which is equivalent to the height of four 40-ft containers. This configuration ensured that the total column height, including the packing, distributor, filler support, and upper and lower clearances, was approximately 10 m. The absorber and regenerator diameters were then determined using the column-sizing function in Aspen HYSYS, assuming a specified packing type and design at approximately 70% of the flooding velocity, while maintaining 90% CO2-removal efficiency.
As illustrated in Figure 2, the capture efficiency in a process utilizing 30 wt% MEA solvent significantly increased with an increase in the L/G ratio. However, beyond a certain threshold, the rate of efficiency improvement diminished, whereas the energy consumption, such as the reboiler duty of the regeneration tower, continued to increase. This trend has been confirmed by previous studies, indicating a clear tradeoff between capture efficiency and energy consumption [5][7][13]. In this study, the L/G ratio was set at 3.5 to balance the efficiency and energy consumption.
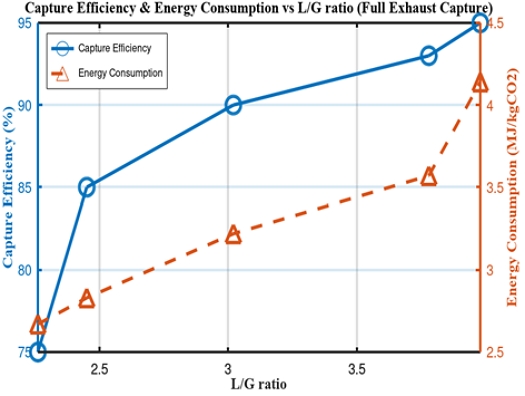
Variation of capture efficiency and energy consumption with L/G ratio under full-exhaust capture scenario
As shown in Table 3, the total exhaust-gas flow rate was 135,900 kg/h, and the absorption and regeneration towers had diameters of 3.38 m and 2.42 m, respectively. The reboiler duty was approximately 3.4 MJ/kgCO2, and the CO2-capture capacity was 7,973 kg/h. Based on these results, assuming a seven-day operational period for a container ship, the vessel would need to store 1,344 tonnes of CO2, necessitating a storage-tank capacity of at least 1,700 m3 at 14 bar. A conversion to ISO tanks shows that this would require loading 70 20-tonne ISO tank containers, imposing a substantial burden on the ship because of its limited space and payload capacity. Consequently, although total exhaust-gas capture offers the technical advantage of a high capture efficiency, it is unfeasible in maritime operating environments because of spatial and economic constraints [7].
3.2 Partial-Exhaust Gas-Absorption Scenario
In the partial-exhaust gas-absorption scenario, a capture capacity of 2 tonCO2/h was conceptually set as a practically installable level for this ship, referring to the 2030 IMO CII reduction requirement, rather than a route-specific CII calculation. According to Clarkson Research, this level is deemed sufficient to maintain CII standards until 2030 [8][9]. As depicted in Figure 3, the partial-capture scenario achieved a stable 90% efficiency within the L/G ratio range of 2.5, with a reboiler duty of approximately 2.9 MJ/kgCO2. This represents a reduction of approximately 15% in the specific reboiler duty per unit of CO2 captured compared to full-exhaust gas capture, thereby demonstrating favorable economic and feasibility outcomes [7].
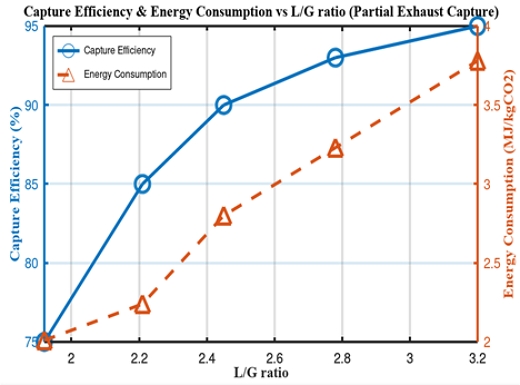
Variation of capture efficiency and energy consumption with the L/G ratio under partial-exhaust capture scenario
The design results for the partial-exhaust gas-absorption scenario are presented in Table 4 as the relative changes with respect to the full-exhaust gas-capture scenario presented in Table 3. Although the same MEA-based process was employed for both full- and partial-exhaust gas capture, significant differences were observed in equipment size, energy consumption, and storage-tank size. The full-capture absorption and regeneration towers were larger, with diameters of 3.38 m and 2.42 m, respectively, complicating their installation on ships. In contrast, the partial-capture towers measured 2.07 m and 1.94 m, respectively, reducing the spatial burden by approximately 30% compared to the full-capture scenario. Furthermore, for a seven-day voyage, the full-capture scenario required approximately 1,700 m3 of LCO2 storage (equivalent to approximately 70 ISO tanks with a 20-tonne capacity), whereas the partial-capture scenario required only approximately 425 m3 (approximately 18 such ISO tanks), which significantly improved the feasibility under shipboard-space and weight constraints.
Relative changes in key design parameters of the partial-capture scenario relative to the full-capture case
From an energy perspective, although full capture offered a higher capture efficiency, the increased reboiler duty necessitated additional energy from the ship. This, in turn, diminished fuel efficiency and directly affected EEDI and CII indices, potentially hindering regulatory compliance. Partial capture, with a reboiler duty of 2.8 MJ/kgCO2, provided a relatively higher energy efficiency (approximately 15%) than full capture, thereby demonstrating superior ship economics. This aligned with the conclusion that the partial-capture scenario offered high economic feasibility during the initial application stage [7].
Therefore, although a full-exhaust gas-capture scenario is ideal, it presents significant challenges for practical applications. Conversely, a partial-exhaust gas-capture scenario is considered a realistic and suitable strategy for phased implementation. The results of this study underscore the necessity for a phased technological-development approach that prioritizes partial capture while ultimately aiming for full-exhaust gas capture when designing future marine OCCS.
These numerical trends are broadly consistent with previous studies on MEA-based CO2 capture for both marine and stationary applications. Luo and Wang [5] and Visoná et al.[6] reported specific reboiler duties of the same order of magnitude as those obtained in this work for high-efficiency MEA based capture and emphasized the strong coupling between L/G ratio, column size, and energy demand. Jung and Seo [10] and Bui et al. [7] likewise showed that moving the full-exhaust to partial-capture configurations, or operating at derated loads, can reduce specific energy consumption while alleviating constraints on column height and Co2 storage capacity. Our full and partial-capture results therefore fall within the ranges reported in the literature and reinforce the conclusion that partial-exhaust gas capture provides a more practical pathway for early-stage deployment of marine OCCS under stringent space and energy limitations.
4. Conclusions
In this study, a carbon-dioxide-capture process using an amine-based solvent was designed for a container ship, and full- and partial-exhaust gas-absorption scenarios were compared and analyzed. The results are summarized as follows:
- 1. In the full-exhaust gas-absorption scenario, a theoretically high capture efficiency of over 90% was achieved. However, the absorption tower (diameter of approximately 3.38 m) and regeneration tower (diameter of approximately 2.42 m) were enlarged, and the reboiler duty increased to approximately 3.4 MJ/kgCO2, resulting in significant energy consumption. Furthermore, assuming a seven-day operation, approximately 1,344 tonnes of CO2 would require storage, necessitating a storage-tank capacity of over 1,700 m3. These results demonstrate the low feasibility of full-exhaust gas capture in ship environments.
- 2. In the partial-exhaust gas-absorption scenario, the capture rate was designed to be 2 tonCO2/h to meet the IMO regulations. The simulation results indicated that a stable capture efficiency of 90% was achieved within an L/G ratio range of 2.5, and the reboiler duty was reduced to approximately 2.9 MJ/kgCO2. This represents an approximately 15% lower specific reboiler duty per unit of CO2 captured compared with full-exhaust gas capture, demonstrating favorable results in terms of ship-operation economy and energy efficiency. Furthermore, the required storage-tank capacity was approximately 425 m3, which is approximately 25% of the full-capture requirement, significantly reducing the space burden when applied to ships.
- 3. Comprehensive exhaust-gas capture presents substantial potential for reducing greenhouse-gas emissions; however, its implementation during the initial commercialization phase is constrained by the limited space on ships and high energy demands. In contrast, partial exhaust-gas capture is deemed the most suitable strategy for the initial deployment of shipboard OCCS, given the need to balance the capture efficiency with energy consumption.
- 4. This study illustrates the feasibility of designing a shipboard OCCS with the long-term objective of full capture while initially adopting partial capture. This approach necessitates a phased technological roadmap that initially achieves regulatory compliance through partial capture, and subsequently progresses to full capture through technological advancements and enhancements in shipboard CO2 storage and reliquefaction systems. In future work, the OCCS performance will be coupled with realistic annual operating profiles to quantify the resulting CII rating improvement for this ship, and a comprehensive energy assessment, including CO2 compression and liquefaction, will be performed to evaluate the total ship-level energy penalty.
Acknowledgments
This research was supported by Korea Institute of Marin Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (RS-2025-00256331).
Author Contributions
Conceptualization, S. T. Jung and Y. S. Choi; Methodology, S. T. Jung; Software, S. T. Jung; Validation, S. T. Jung and T . W. Lim; Formal Analysis, S. T. Jung; Investigation, S. T. Jung; Resources, S. T. Jung; Data Curation, S. T. Jung; Writing—Original Draft Preparation, S. T. Jung; Writing—Review & Editing, T. W. Lim and Y. S. Choi; Visualization, S. T. Jung; Supervision, Y. S. Choi; Project Administration, Y. S. Choi; Funding Acquisition, Y. S. Choi.
References
- International Maritime Organization (IMO), Initial IMO strategy on reduction of GHG emissions from ships (MEPC.304(72)), London, U.K.: IMO, 2018.
- International Maritime Organization (IMO), 2023 IMO strategy on reduction of GHG emissions from ships, London, U.K.: IMO, 2023.
- International Maritime Organization (IMO), Marine Environment Protection Committee (MEPC 80), 3–7 July 2023: official meeting summary page, London, U.K.: IMO, 2023.
-
E. A. Bouman, E. Lindstad, A. I. Rialland, and A. H. Strømman, “State-of-the-art technologies, measures, and potential for reducing GHG emissions from shipping—a review,” Transportation Research Part D: Transport and Environment, vol. 52, pp. 408-421, 2017.
[https://doi.org/10.1016/j.trd.2017.03.022]

-
X. Luo and M. Wang, “Study of solvent-based carbon capture for cargo ships through process modelling and simulation,” Applied Energy, vol. 195, pp. 402-413, 2017.
[https://doi.org/10.1016/j.apenergy.2017.03.027]

-
M. Visonà, F. Bezzo, and F. d’Amore, “Techno-economic analysis of onboard CO₂ capture for ultra-large container ships,” Chemical Engineering Journal, vol. 485, 2024.
[https://doi.org/10.1016/j.cej.2024.149982]

-
M. Bui, C. S. Adjiman, A. Bardow, E. J. Anthony, A. Boston, S. Brown, P. S. Fennell, S. Fuss, A. Galindo, L. A. Hackett, J. P. Hallett, H. J. Herzog, G. Jackson, J. Kemper, S. Krevor, G. C. Maitland, M. Matuszewski, I. S. Metcalfe, C. Petit, G. Puxty, J. Reimer, D. M. Reiner, E. S. Rubin, S. A. Scott, N. Shah, B. Smit, J. P. M. Trusler, P. Webley, J. Wilcox, and N. Mac Dowell, “Carbon capture and storage (CCS): The way forward,” Energy & Environmental Science, vol. 11, no. 5, pp. 1062-1176, 2018.
[https://doi.org/10.1039/C7EE02342A]

- DNV GL, Maritime Forecast to 2050 – Energy Transition Outlook 2019, Høvik, Norway: DNV GL, 2019.
-
H. N. Psaraftis, “Decarbonization of maritime transport: To be or not to be?,” Maritime Economics & Logistics, vol. 21, no. 3, pp. 353–371, 2019.
[https://doi.org/10.1057/s41278-018-0098-8]

-
J. Jung and Y. Seo, “Onboard CO₂ capture process design using rigorous rate-based model,” Journal of Ocean Engineering and Technology, vol. 36, no. 3, pp. 168-180, 2022.
[https://doi.org/10.26748/KSOE.2022.006]

-
A. Padurean, A. M. Cormos, and P. S. Agachi, “Pre-combustion CO ₂ capture by gas–liquid absorption for IGCC power plants,” International Journal of Greenhouse Gas Control, vol. 7, no. 4, pp. 1-11, 2012.
[https://doi.org/10.1016/j.ijggc.2011.12.007]

-
I. M. Bernhardsen and H. K. Knuutila, “A review of potential amine solvents for CO ₂ absorption process: Absorption capacity, cyclic capacity and pKa,” International Journal of Greenhouse Gas Control, vol. 61, pp. 27-48, 2017.
[https://doi.org/10.1016/j.ijggc.2017.03.021]

- Aspen Technology, Inc., Acid Gas Cleaning Using Amine Solvents: Validation with Experimental and Plant Data, Bedford, MA, USA: AspenTech, 2019.
-
Z. Zhao, Y. Zhang, Y. Shao, C. Liu, W. Li, H. Fan, H. Dai, Y. Yang, C. Zheng, and X. Gao, “Simulation of SO2 removal process from marine exhaust gas by hybrid exhaust gas cleaning systems (EGCS) using seawater and magnesium-based absorbent,” Separation and Purification Technology, vol. 287, p. 120557, 2022.
[https://doi.org/10.1016/j.seppur.2022.120557]

-
H. Winnes, E. Fridell, and J. Moldanová, “Effects of marine exhaust gas scrubbers on gas and particle emissions,” Journal of Marine Science and Engineering, vol. 8, p. 299, 2020.
[https://doi.org/10.3390/jmse8040299]

-
R. Dugas and G. T. Rochelle, “Absorption and desorption rates of CO₂ with MEA and PZ,” Energy Procedia, vol. 1, no. 1, pp. 1163–1169, 2009.
[https://doi.org/10.1016/j.egypro.2009.01.153]

-
M. Caplow, “Kinetics of carbamate formation and breakdown,” Journal of the American Chemical Society, vol. 90, no. 24, pp. 6795–6803, 1968.
[https://doi.org/10.1021/ja01026a041]

-
R. B. Said, J. M. Kolle, K. Essalah, B. Tangour, and A. Sayari, “A unified approach to CO ₂ –amine reaction mechanisms,” ACS Omega, vol. 5, no. 40, pp. 26125-26133, 2020.
[https://doi.org/10.1021/acsomega.0c03727]

-
A. H. Al-Mamoori, A. Krishnamurthy, A. A. Rownaghi, and F. Rezaei, “Carbon capture and utilization update,” Energy Technology, vol. 5, no. 6, pp. 834–849, 2017.
[https://doi.org/10.1002/ente.201600747]